
batteries
Lithium-Iron Phosphate Battery
Process Solution
For LFP, Iron phosphate source has to be added. Depending on the required properties, some additives are added, especially for LFP, due to its low electric conductivity, carbon source must be included for example, sucrose.
Characteristics of LFP
LFP has high structural and thermal stability because of olivine structure.
So it guarantee intrinsically safe. And LFP is abundant and environment friendly.
However, there are some disadvantages. LFP has low energy density limited by low the voltage and poor rate capability limited by the 1D ionic and poor intrinsic electronic conductivity.
The solution to solve these problems is size reduction to nanometer range with bead Mill such as ZETA, DISCUS, etc.
Size reduction can improve kinetic problems. (diffusion of Li+ within the particles itself)
Because Ionic diffusion constant depends on particle size. To sustain fast charge and discharging in the battery, it is essential to have fast transportation of electrons and lithium ions. When the particle size of LFP becomes small down to nano or sub-micron range, a large proportional of carbon additives is required to connect all active materials.
LFP (Lithium Iron Phosphate) process
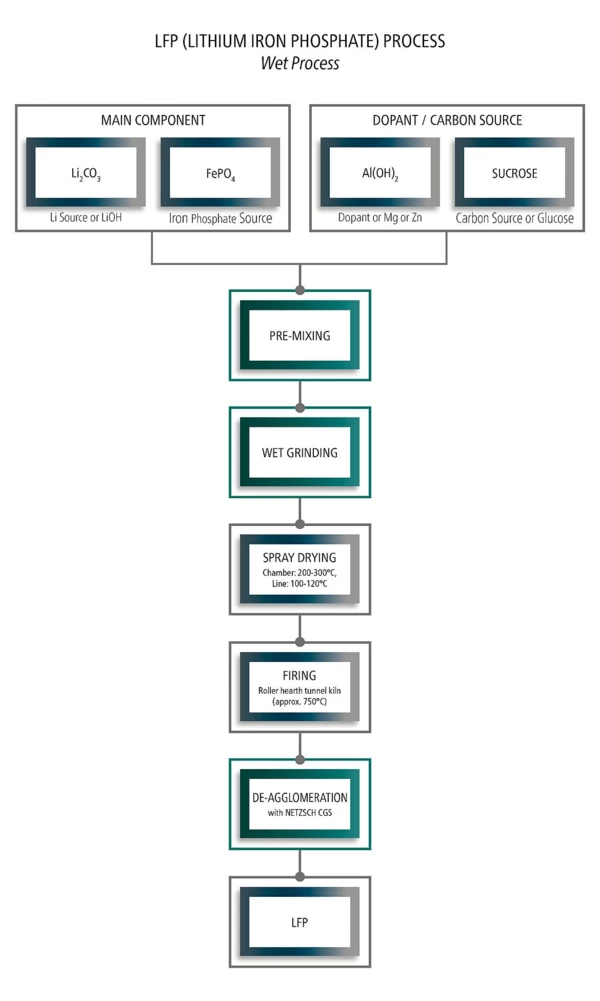
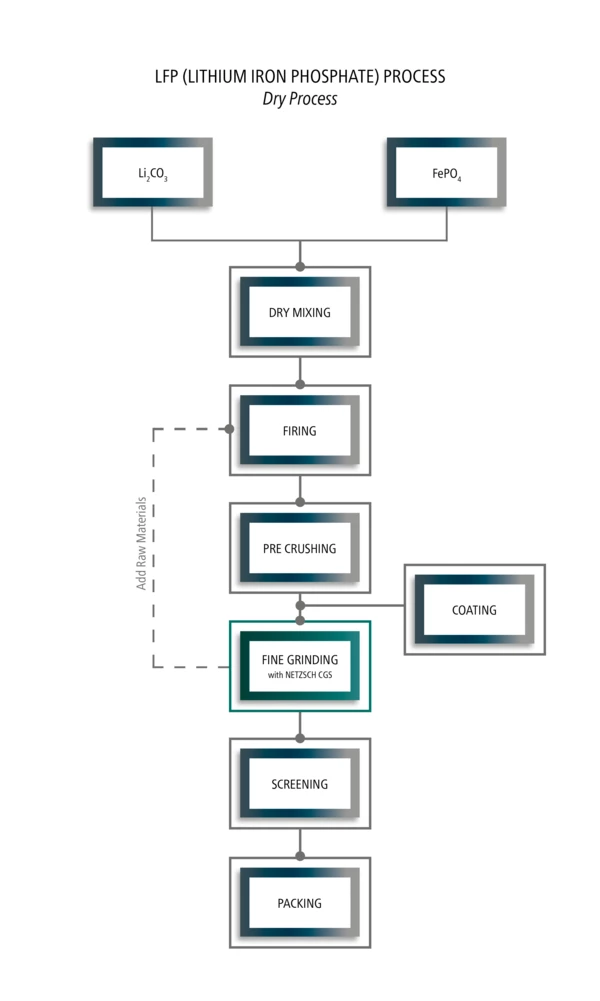
Analyzing Solution
Unlike Lithium-ion batteries, Lithium Iron phosphate batteries (LFP Batteries) are composed of lithium, phosphoric acid, and iron.
Unlike nickel and cobalt materials, phosphoric acid and iron materials have benefits in terms of price, so this is one of the batteries that have been actively researched and developed. However, the key is to increase the energy density of LFP batteries because of the disadvantage of low energy density.
NETZSCH Analyzing Solution provides a solution to energy efficiency.